
HOW TO CHOOSE BACKCOUNTRY SKIS
Pick the best planks for your terrain of choice.
Add $99.00 more to quality for free Shipping!
$0.00 USD

Dear BD,
I recently got into indoor climbing about a year ago and have used my BD harness approximately 50-70 times since then. Yesterday I went top-roping at my local gym and as I was being lowered, I heard a pop. I looked down to see that my harness has ripped off in the waist. I felt myself falling backwards so immediately grabbed the rope and was lowered safely to the ground. I have kept the harness in pristine condition in my gym bag and it has never been abused or used outdoors.
This is a paraphrase from a portion of an email I recently received. Sure I get emails that cause concern, but this was a new one entirely—what did “ripped off in the waist” mean? Was the harness not buckled? Did the webbing cut? Was it pilot error? Was there some sort of manufacturing error? I immediately contacted the customer and asked for some photos, as well as traceability information from the tags on the harness. From this info I was able to tell exactly when the harness was made, the traceability to all raw materials and manufacturing processes and cross reference raw material and finished goods batch testing data. All records and testing results showed nothing out of the ordinary. Below is the photo the customer sent us.
Sure enough, “ripped off at the waist” was an accurate description. It appeared that the waistbelt webbing had somehow cleanly detached from the waist of the harness. It’s worth noting that nowadays most harnesses no longer consist of one continuous piece of structural webbing. Often the webbing used to attach the buckle is connected to a different, usually wider, thinner, lighter, waist material, and all of this is covered with a comfortable and durable shell material. It is at this junction where this harness failed. But how and why? The webbing appeared to be perforated from the needle during the sewing operation, but there was no hint of any thread left. It had to be sewn at one point as he’d used the harness for 70 days without incident. There appeared to be some pink coloring showing on the webbing, but the thread used on that particular harness should have been grey. We asked the customer to send it in so we could take a closer look.

Wow. This ranks up there as one of the weirdest issues we’ve seen. Where the harness broke, it literally looked like the nylon thread had simply melted away. The remaining polyester webbing of the waistbelt was left perforated with remnants of pink coloring. The nylon belay loop had a pink tinge, and the belay loop’s nylon tacks were also pink. In some areas of the harness, the nylon thread tacks were still grey, whereas in other areas, the thread, and only the thread, had turned pink, but was still intact. I grabbed the harness at the visually intact but pink tacks and EASILY pulled them apart by hand. The pink thread had almost no structural integrity. It appeared that the pink was a true indicator of some kind of contamination. As well, the aluminum buckle was showing visible signs of corrosion. It was time to get all CSI (Climbing Scene Investigation).
Our first thought was chemical contamination. The fact that it was only on portions of the harness, there were some splatter marks and some material fading—this made us think that the harness was soaked in some sort of liquid, or something was spilled on it. But it had to be pretty potent to eat the nylon threads like it did.
It was super interesting that the contamination appeared to mainly affect the nylon thread, but didn’t affect the polyester waistbelt webbing of the harness. So what chemicals affected nylon, but not polyester?
Luckily Kevin, one of the crack crew of Quality Engineering guys here has a background in chemical engineering. He started throwing out terms like molar, reagent, polymer and covalent bonds. I cut him loose on investigating different chemicals and their effect on nylons and polyesters.
With a bit of internet research and Kevin’s initial analysis, all signs pointed towards hydrochloric or sulphuric acid. With a bit more digging, Kevin found lists of common household items that contain different levels of these acids.

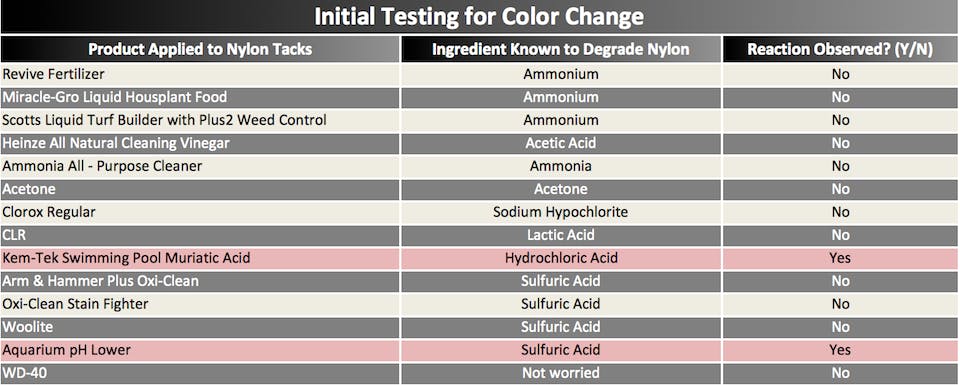
After a few trips to the grocery store, hardware store & a pet shop, Kevin had a makeshift chem lab set up. Of course we couldn’t test everything, but we tried to choose chemicals with the highest concentration of acid giving us a higher probability of seeing some sort of reaction. The goal was to apply these chemicals to similar harnesses and:
a) see if the nylon thread turned pink (our believed indicator) while not affecting the polyester webbing
b) determine if they weakened the structural integrity to a point where it would fail at extremely low loads.
In the meantime we were in contact with the customer on several occasions, trying to get a more complete picture of the history of the harness—how it was used, where it was stored, any opportunities for chemical contamination. He said it was always in his gym bag either in the trunk of his car or in his house, or of course wearing it at the gym. It had never been left anywhere and he had never worn it outside. Sure there were some instances where there were some groceries and cleaning supplies, etc. in the trunk at the same time, but nothing that noticeably leaked, and likely nothing with an acid concentration high enough to attack the nylon threads.
While Kevin was performing his comparative chemical experiments, we sent the customer’s actual harness off to a lab that specializes in analyzing chemical composition and contamination. We were hoping they could narrow down the contaminant and suggest possible common items with a concentration of acid high enough that it could affect nylon as we saw.

Ah ha. A smoking gun or guns? Out of all the different chemicals that we applied to the harnesses, two turned the nylon tacks instantly pink—muriatic acid, which contains 14.5% hydrochloric acid, and pH Adjuster, which is used to clean aquariums, and contains varying amounts of sulphuric acid, depending on how you mix it. The sample harnesses with these two chemicals looked almost identical to the customer’s contaminated harness.

With excessive exposure (ie. submersion for extended time) to these chemicals, we could recreate the level of degradation of the nylon threads and easily pull the tacks apart by hand just as we saw with the customer’s harness.
Given our testing we were pretty sure that the contamination event had to happen just a few days prior to the incident. Hopefully this could help the customer narrow down his search for possible opportunities for contamination.
So re-creating the failure was one thing—we were pretty confident that the harness was contaminated by either sulphuric acid or hydrocholoric acid—but we still didn’t know how the customer’s harness was exposed to these chemicals.
More research and phone calls:
• Muriatic acid used to be used to clean holds in climbing gyms. We contacted several gyms and a pro route-setter that we know that has worked in pretty much every gym in the West in the last 20 years, and apparently no one has been using muriatic acid to clean gym holds for years. The customer said he’s never been in proximity to cleaning holds, moving a bucket of holds, etc.
• I learned that muriatic acid is a key ingredient when cooking meth. The customer doesn’t cook meth—I asked him and he wasn’t psyched about it.
• Muriatic acid is used to clean concrete and related tools. No chance of exposure there says the customer.
• Sulphuric acid is most common in car batteries. The customer had a BRAND NEW car battery in the trunk of his car, perhaps with his harness, but only for a few minutes, there was no apparent leakage, and this was several months before the incident.
• Sulphuric acid is in a chemical that is commonly used to clean aquariums (pH Adjuster). The customer doesn’t have an aquarium.
So, though we thought we had it narrowed down to one of two chemicals, we were at a bit of a standstill as to how this particular harness actually got contaminated.
We received the lab report back, along with some cool photos taken with a Scanning Electron Microscope and Infrared Spectroscopy—the test showed traces of sulphur on the contaminated pink thread and the corroded buckle. You can see by looking at a few of these photos that the affected pink thread looks to be eaten or melted by the acid, whereas the non-affected gray thread is more uniform. As well, the traces of sulphur on the aluminum buckle is visually obvious.
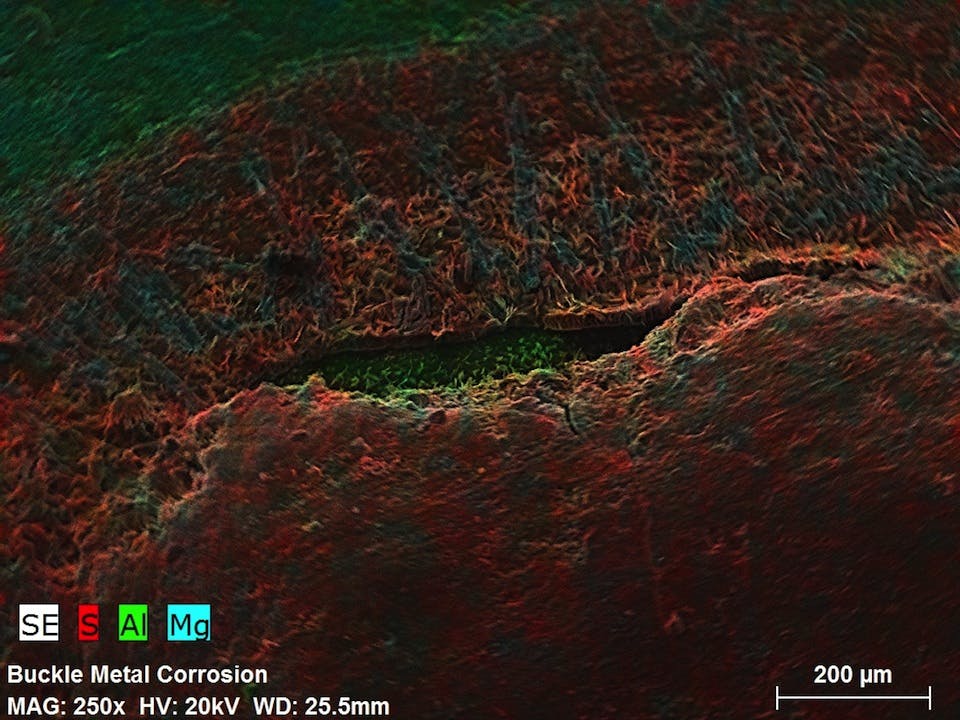
So contamination by sulphuric acid is what it looks like, but still no real explanation of how.
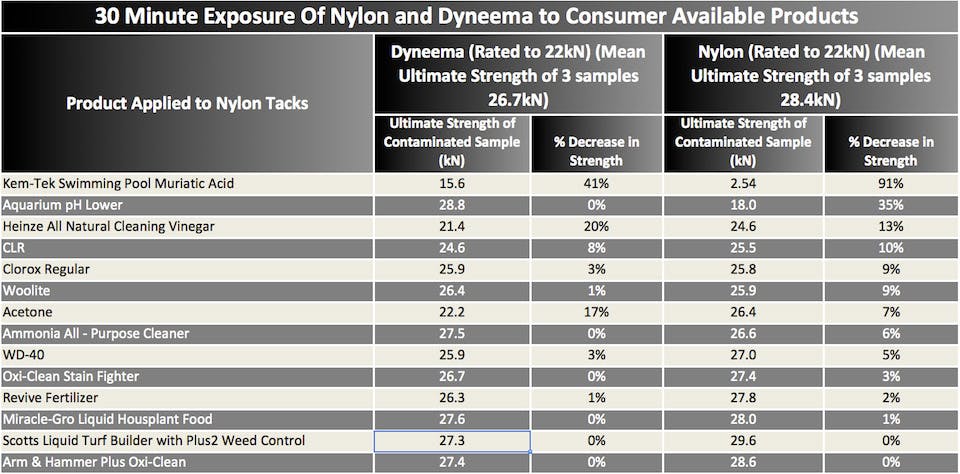
Seeing as we had all of these chemicals around, and had learned a bit about certain chemicals affecting nylon and not polyester, we decided to do a controlled experiment to see if, and by how much, these chemicals affect nylon slings and, while we’re at it, Dyneema. The point was hopefully to shed some light on some common questions we get like, ‘I got some acetone on some slings,’ or ‘I accidentaly got some WD-40 on my cam slings while cleaning them – do I need to be worried?’.
Once again—no massive, PhD-level scientific experiment here, just one sample of each:
First round: each sample soaked in the chemical for 30 minutes, dried for 15 minutes, and was then tested to failure. Based on these results, we picked the chemicals that showed some significant signs of degradation to the materials and did another more intense round of experiments.
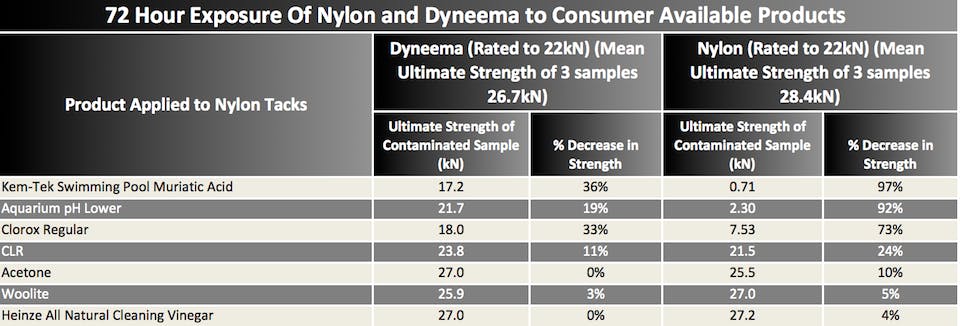
Second round: each sample soaked in the chemical for 72 hours, dried for 24 hours, and was then tested to failure.
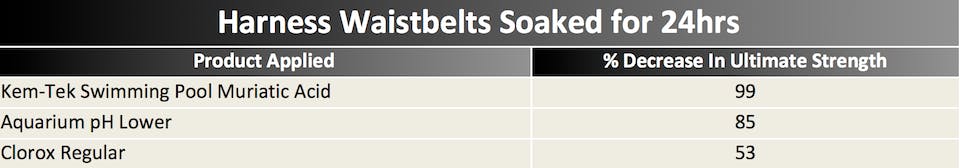
It’s difficult to make any concrete conclusions as we tested only one of each sample, but it’s obvious that the chemicals that affected the strength of the materials the most were:
1) muriatic acid (basically hydrochloric acid)
2) aquarium cleaner (pH adjuster, contains sulphuric acid)
3) bleach
Now that we had validated both muriatic and sulphuric acids as having a catastrophic effects on materials as well as being a match for the change in thread color, we chose to test them on the actual structural integrity of a harness. Kevin contaminated portions of a few full harnesses and tested them to failure to see just how they would perform.

We learned that the gray nylon thread turns pink when contaminated with acid. Why exactly this is at the molecular level we didn’t dig too deeply – but it was cool, and ended up being a great indicator in this case which really helped focus the investigation quickly to acids.
We were able to re-create the visual and structural degradation of the customer’s harness by exposing similar harnesses to sulphuric acid. And therefore though we do believe the customer’s harness to have been contaminated by some sulphuric acid, unfortunately he/we could not determine exactly which chemical or how it became exposed.

Further testing showed that as one would think, acids had a much greater detrimental affect on the nylons and Dyneema than the other chemicals tested.
As well the duration of exposure played a role in strength reduction of nylon and Dyneema, especially when looking at the acids.
As usual, there is much more testing we could perform:
- Further investigation into if and what colors different threads changed when exposed to acids and why
- More in-depth investigation into why the acid affects nylon and not polyester
- Testing of additional chemicals on nylon and Dyneema
Several years ago there was a rope incident and after analysis it ended up looking like the rope was somehow contaminated with acid. How the rope actually came in contact was never determined. So this is the second incident I’ve personally looked into regarding chemical contamination of climbing gear – and luckily in both cases no one was hurt. Also in both cases, the end result was that the customer doesn’t really know how this chemical contamination occurred. Scary.
The fact that the customer and our team were never able to determine the exact cause of contamination of his harness is the whole point—protecting your gear from hazardous materials and visually inspecting it on a regular basis is of the utmost importance.
All manufacturers say it in the instructions multiple times. Don’t give your gear the opportunity to be contaminated. Store your gear properly, use rope bags, and know the complete history of your gear. If you’re unsure if your gear may have been exposed to harmful chemicals, retire it. Likewise, buying used textile climbing equipment is never recommended.
A decent general rule of thumb that I’ve been told by some rope guru friends of mine “if it’s ok to put on your skin, it’s probably not going to damage nylons and other textiles.” Of course you can never be sure—when it doubt always ask the manufacturer if they happen to have any knowledge of if and how a particular chemical can affect the integrity of a piece of climbing gear. And if you’re really in doubt, it’s always best to play the conservative card and retire the gear.
Be safe out there,
KP


Inspired by climbing, created by hand, our Spring 2026 prints tell a deeper story.

Basic field maintenance and repair tips for your climbing skins from our Reroute team.

Watch Connor take down another classic testpiece on the Empath cliff in Kirkwood, California.






Follow BD Athlete Yannick Glatthard deep into the Swiss Alps as he shares his home...
Follow BD Athlete Yannick Glatthard deep into the Swiss Alps as he shares his home mountains with close friends.


Follow Dorian Densmore and Mya Akins for another winter season of steep Alaskan spines, backyard...
Follow Dorian Densmore and Mya Akins for another winter season of steep Alaskan spines, backyard couloirs, and deep adventures in the mountains.


Watch BD Athlete Alex Honnold throw down on some hard trad high above Tahoe.

